The pharmaceutical industry is currently grappling with a paradox where the exponential growth of biological data has not yet translated into a proportional increase in drug approval rates. While artificial intelligence was promised as the ultimate solution to this stagnation, the reality of implementation has been hampered by rigid data silos and the immense logistical burden of maintaining high-performance local computing clusters. Federated learning has emerged as a transformative architectural shift, allowing institutions to train powerful predictive models on distributed datasets without ever moving the sensitive raw information from its original source. This review examines how this decentralized approach, combined with modern cloud integration, is reshaping the competitive landscape of molecular modeling.
Introduction to Collaborative AI in Drug Development
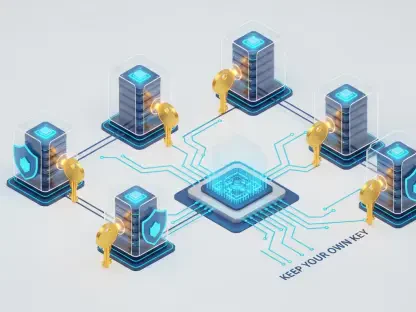
The core principle of federated learning in this sector involves a radical departure from the traditional centralized data warehouse model. Historically, a researcher needing to train a deep learning model for protein-ligand binding would require a massive, unified dataset, which often meant months of legal negotiations and data cleaning across different institutions. In the current landscape, the technology functions by bringing the code to the data rather than the data to the code. This mechanism allows a central server to orchestrate training across multiple independent nodes—such as private biotech databases or hospital records—collecting only the mathematical updates to the model weights.
This evolution is significant because it addresses the “data scarcity” problem that plagues even the largest global pharmaceutical companies. No single entity possesses a complete view of the chemical universe; one firm may have extensive data on small molecules while another excels in macrocycles. By utilizing a federated framework, these organizations can benefit from a collective intelligence that is statistically more robust and generalizable than any siloed model. This shift moves the industry away from isolated experimentation toward a cooperative computational ecosystem where the strength of the algorithm is derived from the breadth of the network.
Core Architectural Components of Modern AI Pipelines
Models-as-a-Service (MaaS) and Cloud Integration
The transition toward Models-as-a-Service (MaaS) represents a critical operational upgrade that removes the “it-infrastructure tax” previously paid by research teams. In this setup, sophisticated machine learning models are delivered via cloud-native platforms like Signals Xynthetica, which treat the model not as a static file, but as a live, evolving service. This architecture matters because it decouples the scientific inquiry from the underlying hardware requirements. Scientists can now trigger complex simulations directly from their electronic lab notebooks, with the MaaS layer handling the heavy lifting of version control, environment scaling, and secure API management.
This integration is unique because it embeds predictive power directly into the daily laboratory workflow. Instead of a chemist having to export data, send it to a specialized data science team, and wait days for a response, the cloud-integrated model provides real-time feedback on molecular properties. This immediacy changes the nature of hypothesis testing, allowing for rapid iterations that were previously impossible due to technical friction. The “service” aspect ensures that the models are constantly tuned and updated against the latest global benchmarks, preventing the performance decay that typically affects locally hosted algorithms.
Decentralized Data Processing and Aggregation
Decentralization is the technical backbone that makes large-scale collaboration palatable to risk-averse legal departments. By utilizing decentralized data processing, the raw intellectual property—the specific chemical structures and experimental results—remains behind the participant’s firewall. Only the “delta,” or the change in the model’s parameters after seeing the local data, is shared with the central aggregator. This ensures that even if the central server were compromised, no proprietary chemical secrets would be revealed, as the aggregated weights are abstract mathematical values that cannot be easily reversed into their original data points.
This implementation is distinct from traditional anonymization techniques, which are often prone to re-identification attacks. Federated aggregation uses secure multi-party computation and differential privacy to add layers of mathematical noise, ensuring that the contribution of any single data point is hidden within the group. For the industry, this means that the competitive advantage is no longer just about who has the most data, but who can best leverage the collective insights of the network. It levels the playing field, allowing smaller biotech startups with high-quality, niche data to contribute to and benefit from models trained on the vast historical archives of industry giants.
Emerging Trends in Precompetitive Research
A notable shift is occurring in how the industry views “precompetitive” space, with more companies realizing that general molecular physics and ADMET (Absorption, Distribution, Metabolism, Excretion, and Toxicity) profiles are better solved collectively. Innovations are now focusing on hybrid models that combine physics-based simulations with these federated machine learning insights. This trend is driven by the realization that while the specific drug target is a proprietary secret, the way a molecule interacts with a standard human liver enzyme is a universal challenge that everyone benefits from solving accurately.
Furthermore, there is a growing movement toward “transfer learning” within these federated networks. This involves taking a model trained on a broad, multi-partner dataset and “fine-tuning” it on a very small, highly specific internal dataset. This approach allows a company to maintain a unique edge in a specific therapeutic area while utilizing a foundation built on the collective wisdom of the entire network. Such developments indicate a move toward more modular AI, where components can be swapped and optimized for specific biological contexts without rebuilding the entire system from scratch.
Real-World Applications and Industrial Implementations
The most impactful application of this technology is currently seen in the expansion of chemical libraries for neglected diseases and rare conditions. In these fields, data is notoriously sparse, making traditional AI approaches ineffective. By using a federated approach, international consortia are pooling disparate clinical and chemical data points to identify promising candidates that would have been missed in smaller trials. For example, the collaborative efforts between major players like Eli Lilly and specialized biotech firms demonstrate that even competitors can find common ground when the goal is to map the foundational “rules” of molecular behavior.
Another significant implementation is in the optimization of lead compounds during the medicinal chemistry phase. Systems like Lilly’s TuneLab allow researchers to project how a theoretical change in a molecule’s structure might affect its efficacy by drawing on the patterns learned from millions of previous experiments conducted across the network. This doesn’t just speed up the process; it reduces the number of “dead-end” molecules synthesized in the lab, saving millions of dollars in reagents and personnel time. The real-world value lies in this reduction of physical trial-and-error, shifting the cost curve of drug development downward.
Technical Barriers and Intellectual Property Concerns
Despite the promise, several technical hurdles remain, particularly regarding “data heterogeneity.” When data comes from different labs, the “noise” or bias introduced by different experimental protocols can confuse the global model. If one lab uses a different assay temperature than another, the federated model might struggle to reconcile the conflicting results. Current development efforts are focused on creating automated “data harmonization” layers that can normalize these differences before the training updates are sent to the aggregator. This is a critical step for ensuring that the global model remains accurate across diverse environments.
Intellectual property concerns also persist, specifically regarding the “ownership” of the final global model. If fifty companies contribute to a model that identifies a multi-billion-dollar drug, the question of who owns that insight remains legally complex. Most current implementations circumvent this by treating the global model as a shared utility—a “precompetitive” tool—while the specific molecules discovered using that tool remain the property of the discoverer. However, as models become more specialized, the industry will need to establish clearer regulatory and legal frameworks to manage the value generated by collaborative AI.
The Future of Globalized Molecular Modeling
The trajectory of this technology points toward a future where “Globalized Molecular Modeling” becomes the standard operating procedure for all drug discovery. We are likely to see the emergence of specialized federated exchanges, where data is not just shared, but tokenized and traded based on its quality and predictive value. This would create a market for high-quality scientific data, incentivizing labs to produce cleaner, more reliable results. Eventually, these decentralized networks could integrate real-world evidence from wearable devices and electronic health records, closing the loop between the chemistry lab and the patient’s bedside.
Breakthroughs in “zero-knowledge proofs” will likely solve the remaining trust issues, allowing participants to prove their data is valid without revealing any of its content. As these technologies mature, the distinction between “computational” and “experimental” drug discovery will blur. We are moving toward a state where every pipette stroke in a lab in Singapore can, within seconds, refine the predictive accuracy of a model being used by a chemist in Boston. This interconnectedness will fundamentally shorten the time required to move from a biological hypothesis to a clinical-stage therapeutic.
Conclusion and Strategic Assessment
The shift toward federated learning and Models-as-a-Service has redefined the boundaries of pharmaceutical innovation by effectively decoupling data utility from data ownership. This review established that the primary value of these systems lies not just in their predictive accuracy, but in their ability to foster a collaborative research environment that respects the stringent privacy requirements of the industry. The integration of these tools into cloud-native platforms has successfully lowered the barrier to entry, allowing scientists to focus on biology rather than the underlying computational plumbing.
The transition to decentralized AI was a strategic necessity in an era where data silos were the primary obstacle to scientific progress. Organizations that adopted these federated frameworks early have gained a significant lead by contributing to and benefiting from a more generalized understanding of molecular space. While technical challenges regarding data standardization remain, the trajectory of the technology suggests that the era of isolated, proprietary-only modeling is coming to an end. The industry has moved toward a more resilient, collective approach that promises to deliver safer and more effective therapies at a pace that was previously unattainable.